
Water Deionization Process
Among all common water filtration methods, the process used for the removal of all dissolved salts from water is referred to as deionization. Deionization requires the flow of water through two ion exchange materials in order to effect the removal of all salt content.
The terms demineralization and deionization are used somewhat interchangeably by the industry. While the term demineralization is generally better understood, deionization is especially apt.
The passage of water through the first exchange material removes the calcium and magnesium ions just as in the normal softening process. Unlike home equipment, deionization units also remove all other positive metallic ions in the process and replace them with hydrogen ions instead of sodium ions.
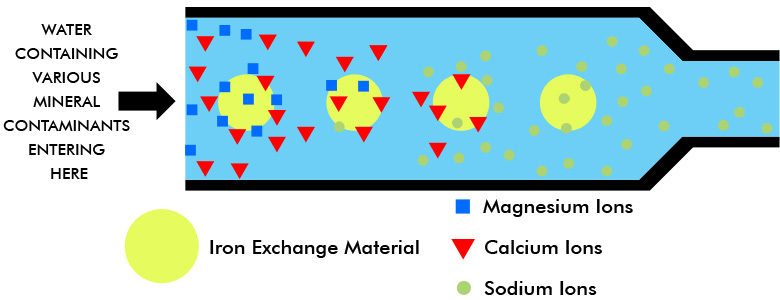
As the metallic ions in the water affix themselves to the exchange material, the latter releases its hydrogen ions on a chemically equivalent basis. A sodium ion (Na+) displaces one hydrogen ion (H+) from the exchanger; a calcium ion (Ca++) displaces two hydrogen ions; a ferric ion (Fe+++) displaces three hydrogen ions, etc. (Recall those home softeners also release two sodium ions for every calcium or magnesium ion they attract.)

