
Different water filtration methods explained (Continued 2)
Reverse Osmosis
Reverse osmosis (RO) is the most economical method of removing 90% to 99% of all contaminants. The pore structure of RO membranes is much tighter than UF membranes. RO membranes are capable of rejecting practically all particles, bacteria and organics >300 daltons molecular weight (including pyrogens). In fact, reverse osmosis technology is used by most leading water bottling plants. Natural osmosis occurs when solutions with two different concentrations are separated by a semi-permeable membrane. Osmotic pressure drives water through the membrane; the water dilutes the more concentrated solution, and the end result is an equilibrium.
In water purification systems, hydraulic pressure is applied to the concentrated solution to counteract the osmotic pressure. Pure water is driven from the concentrated solution and collected downstream of the membrane.
Because RO membranes are very restrictive, they yield slow flow rates. Storage tanks are required to produce an adequate volume in a reasonable amount of time.

RO also involves an ionic exclusion process. The only solvent is allowed to pass through the semi-permeable RO membrane, while virtually all ions and dissolved molecules are retained (including salts and sugars). The semi-permeable membrane rejects salts (ions) by a charge phenomena action: the greater the charge, the greater the rejection. Therefore, the membrane rejects nearly all (>99%) strongly ionized polyvalent ions but only 95% of the weakly ionized monovalent ions like sodium.
Reverse osmosis is highly effective in removing several impurities from water such as total dissolved solids (TDS), turbidity, asbestos, lead and other toxic heavy metals, radium, and many dissolved organics. The process will also remove chlorinated pesticides and most heavier-weight VOCs. Reverse osmosis and activated carbon filtration are complementary processes. Combining them results in the most effective treatment against the broadest range of water impurities and contaminants.
RO is the most economical and efficient method for purifying tap water if the system is properly designed for the feed water conditions and the intended use of the product water. RO is also the optimum pretreatment for reagent-grade water polishing systems.
In addition, Reverse osmosis treatment is an insurance policy against nuclear radiation such as radioactive plutonium or strontium in the drinking water. If one lives near a nuclear power plant, this is a key way to ensure the household is drinking the best water for their health.
| |||||||
Ultraviolet (UV) Radiation
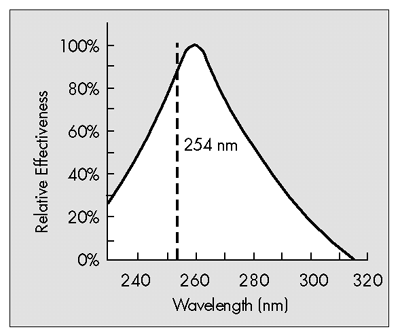
Ultraviolet radiation has widely been used as a germicidal treatment for water. Mercury low-pressure lamps generating 254 nm UV light are an effective means of sanitizing water. The adsorption of UV light by the DNA and proteins in the microbial cell results in the inactivation of the microorganism.
Recent advances in UV lamp technology have resulted in the production of special lamps which generate both 185 nm and 254 nm UV light. This combination of wavelengths is necessary for the photooxidation of organic compounds. With these special lamps, Total Organic Carbon (TOC) levels in high purity water can be reduced to 5 ppb.
| |||||||
Pulling It All Together
Water Purification Systems
Because each purification technology removes a specific type of contaminant, none can be relied upon to remove all contaminants to the levels required for critical applications. A well-designed water purification system uses a combination of purification technologies to achieve final water quality.
Each of the purification technologies must be used in an appropriate sequence to optimize their particular removal capabilities. The schematic below shows a central laboratory water purification system designed to produce water for critical applications.
The first step is pretreatment equipment specifically designed to remove contaminants in the feed water. Pretreatment removes contaminants that may affect purification equipment located downstream, especially reverse osmosis (RO) systems. Examples of pretreatment are: carbon filters (or tanks) for chlorine removal, particulate filters for sediment/silt/particulate removal, and softening agents to remove minerals that cause "hard" water.
The next purification step is Reverse Osmosis (RO). RO removes 90 to 99% of all the contaminants found in water. It is the heart of any well-designed water purification system because it effectively removes a broad range of contaminants.
However, the tight porosity of the RO membrane limits its flow rate. Therefore, a storage container is used to collect water from the system and distribute it to other points-of-use such as polishing systems.
Polishing systems purify pretreated water, such as RO water, by removing trace levels of any residual contaminants. Polishing elevates the quality of pretreated water to "Type I" or "ultrapure" water.
A polishing system is designed to remove residual traces of impurities from water already pretreated by some other means (such as reverse osmosis or deionization). Treating raw tap water using such a system would quickly exhaust its capacity and affect final quality.
A typical polishing system may consist of activated carbon, mixed-bed deionization, organic scavenging mixtures, and 0.22 µm final filtration. Systems can also be enhanced with ultrafiltration, ultraviolet oxidation, or other features for use in specific applications.
This combination of purification technologies, combined with proper pretreatment, will produce water that is virtually free of ionic, organic, and microbial contamination.

